Lynn Margulis famously and controversially championed the theory that [[Symbiosis]], not just random mutation, is a primary driver of evolution, most notably through her serial endosymbiotic theory for the origin of complex cells.
# Evidence for endosymbiosis
https://evolution.berkeley.edu/it-takes-teamwork-how-endosymbiosis-changed-life-on-earth/evidence-for-endosymbiosis/
Biologist Lynn Margulis first made the case for [[endosymbiosis]] in the 1960s, but for many years other biologists were skeptical. Although the microbiologist [[Kwang Jeon]] had watched amoebae become infected with x-bacteria and then evolve to depend upon them, no one was around over a billion years ago to observe the events of endosymbiosis. Why should we think that a mitochondrion used to be a free-living organism in its own right? It turns out that many lines of evidence support this idea. Most important are the many striking similarities between prokaryotes (like bacteria) and mitochondria:
When you look at it this way, mitochondria really resemble tiny bacteria making their livings inside eukaryotic cells! Based on decades of accumulated evidence, the scientific community supports Margulis’s ideas: endosymbiosis is the best explanation for the [[Evolution]] of the eukaryotic cell.
What’s more, the evidence for endosymbiosis applies not only to mitochondria, but to other cellular organelles as well. [Chloroplasts](https://evolution.berkeley.edu/glossary/chloroplast) are like tiny green factories within plant cells that help convert [[Energy]] from sunlight into sugars, and they have many similarities to mitochondria. The evidence suggests that these chloroplast organelles were also once free-living bacteria.
The endosymbiotic event that generated mitochondria must have happened early in the [[History]] of eukaryotes, because all eukaryotes have them. Then, later, a similar event brought chloroplasts into some eukaryotic cells, creating the lineage that led to plants.
[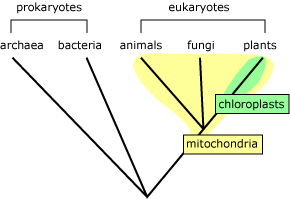](https://evolution.berkeley.edu/wp-content/uploads/2021/08/eukaryote_clade.gif)
Despite their many similarities, mitochondria (and chloroplasts) aren’t free-living bacteria anymore. The first eukaryotic cell evolved more than a billion years ago. Since then, these organelles have become completely dependent on their host cells. For example, many of the key [[Proteins]] needed by the mitochondrion are imported from the rest of the cell. Sometime during their long-standing relationship, the genes that code for these proteins were transferred from the mitochondrion to its host’s genome. Scientists consider this mixing of genomes to be the irreversible step at which the two independent organisms become a single individual.
**Grabbing take-out: _Paramecium bursaria_ packs a lunch**
[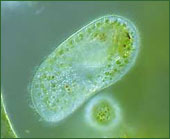](https://evolution.berkeley.edu/wp-content/uploads/2021/08/paramecium.jpg "P. bursaria, photo © www.micrographia.com.")
_P. bursaria_, photo © [www.micrographia.com](http://www.micrographia.com/).
_Paramecium bursaria_, a single-celled eukaryote that swims around in pond water, may not have its own chloroplasts, but it does manage to “borrow” them in a rather unusual way. _P. bursaria_ swallows photosynthetic green algae, but it stores them instead of digesting them. In fact, the normally clear paramecium can pack so many algae into its body that it even looks green! When _P. bursaria_ swims [[into the light]], the algae photosynthesize sugar, and both cells share lunch on the go. But _P. bursaria_ doesn’t exploit its algae. Not only does the agile paramecium chauffeur its algae into well-lit areas, it also shares the food it finds with its algae if they are forced to live in the dark.